Sickle cell disease (SCD) affects millions of people around the globe and is the 4th leading cause of deaths in children in many developing countries. It causes a number of health problems, such as attacks of pain, anaemia, swelling in the hands and feet, bacterial infections and stroke. Sickle-cell contributes to a low life expectancy in the developed world of 40 to 60 years.
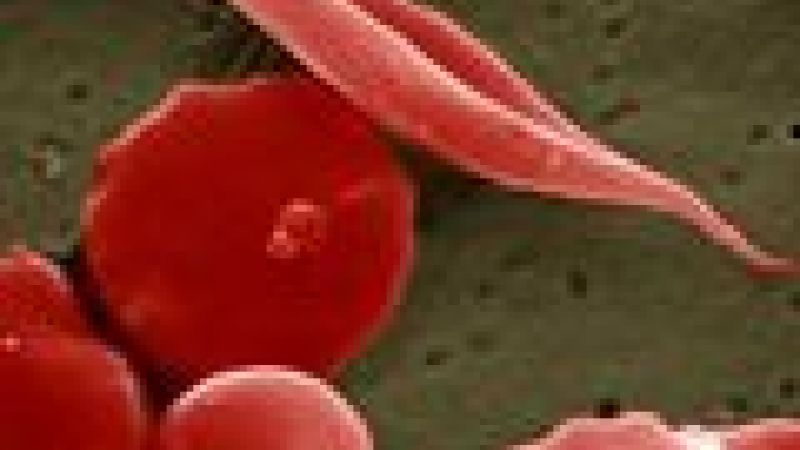
The disease results in abnormal haemoglobin - the oxygen-carrying protein found in red blood cells – giving the blood cell a rigid, sticky, sickle-like shape that hinders its oxygen-binding properties. These irregularly shaped cells can get stuck in small blood vessels, which can slow or block blood flow and oxygen to parts of the body. A blood and bone marrow transplant is currently the only cure for sickle cell disease, but only a small number of patients are eligible. For the rest, there's no cure but effective treatments can relieve pain, help prevent problems associated with the disease and prolong life.
70 years ago, researchers found a genetic connection to the anatomical abnormalities seen in blood cells. A mutation seemed to be causing the moon-shaped blood cells. The most severe form of the disease occurs when two copies of the mutation are inherited. However, patients with one sickle cell gene, referred to as sickle cell trait, usually do not have any of the signs of the disease and live a normal life, but they can pass the trait on to their children.
As with all inherited genetic diseases, you’d expect natural selection to weed out a gene that has such unpleasant consequences but with sickle cell disease, that doesn’t seem to be the case. Indeed, as of 2015, about 4.4 million people have sickle cell disease, while an additional 43 million have sickle cell trait. So what makes the disease stay in the human population?
Researchers found the answer by looking at where the disease was most prevalent. As it turns out, 80% of sickle cell disease cases occur in Sub-Saharan Africa or amongst populations having their ancestors in this region, as well as in other parts of the world where malaria is or was common. There was a long standing theory that the sickle cell trait – having only one sickle cell gene – didn’t cause discomfort and provided a bonus trait of preventing patients from contracting severe forms of malaria. Later confirmed - associating sickle cell to a 29% reduction in malaria incidence - this working theory would explain why the mutation stuck around in evolution. In 2011, researchers used mice to confirm the assumption.
Miguel Soares and Ana Ferreira of the Gulbenkian Institute of Science in Oeiras, Portugal, and colleagues found that haem – a component of haemoglobin – is present in a free form in the blood of mice with sickle cell trait, but largely absent from normal mice. By injecting haem into the blood of normal mice before infecting them with malaria, researchers found it could help guard against malaria. The mice did not develop the disease. Their results also showed that the gene does not protect against infection by the malaria parasite, but prevents the disease taking hold after the animal has been infected.

In another experiment, Soares and his team showed that a build-up of Toxic T-cells after infection with malaria is inhibited in mice with one sickle cell gene. For a long time, scientists assumed that the sickle cell trait enhanced resistance, in other words, people with the variant were better at fighting infection by the malaria parasite.
But it became clear that since humans/animals with the sickle-cell trait have to break down and detoxify misshapen red blood cells their entire lives, their molecular machinery for cleaning up mess is already in place when the malarial parasite attacks them. Therefore, infected sickled red blood cell are actually a hostile environment for the plasmodium parasite.
The sickle cells have membranes, stretched by their unusual shape, that become porous and leak nutrients that the parasites need to survive and the faulty cells eventually get eliminated quite fast by the organisms, destroying the parasite along the way.
These findings help better understand the dynamics at play during an infection with the malaria causing plasmodium parasite and could open the way to new therapeutic targets and options to treat malaria, a disease that continues to inflict tremendous medical, social and economic burdens to a large proportion of the human population.
Read more on Malaria
Fighting the world's most deadly animal: the mosquito
Last edited: 11 July 2023 16:10