 Gene therapy using a mutant form of a gene known to be involved in many of the most common cancers can destroy tumours in mice without any major side effects. The tests in mice also show that the animals do not become resistant to the treatment, even after a year. Because this new research is promising, the researchers are already looking to translate the work into a form that can be tested in patients.
Gene therapy using a mutant form of a gene known to be involved in many of the most common cancers can destroy tumours in mice without any major side effects. The tests in mice also show that the animals do not become resistant to the treatment, even after a year. Because this new research is promising, the researchers are already looking to translate the work into a form that can be tested in patients.
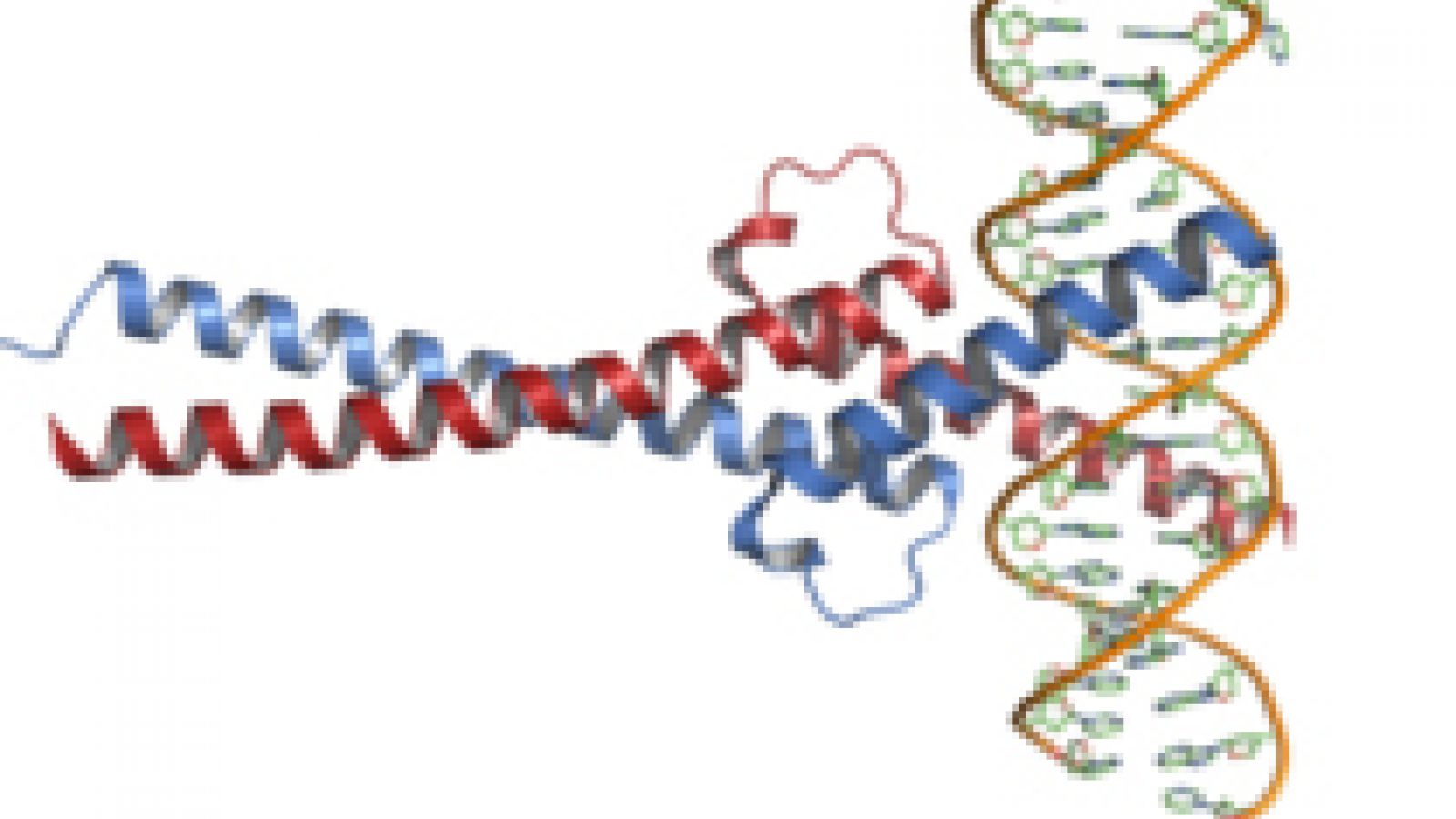
The gene therapy targets a protein called Myc, which has been long known to play a role in the development of many different types of cancer, including cervical, breast, colon, lung and stomach cancer. Myc is important because it controls multiple cellular processes: how fast cells grow, what types of cells they differentiate into, and even cell death. When these go wrong, a tumour is often the disastrous result.
In previous research, the team found that they were able to suppress the activity of Myc using a mutant form of the same protein. The mutant, named Omomyc, binds to Myc and selectively inactivating the protein in key processes linked to tumour formation.
The gene therapy delivers a gene encoding Omomyc into the lung cells of mice. Crucially, the team modified the gene so that it could be turned on and off by a chemical added to the mouse's drinking water.
Mice genetically engineered to develop multiple lung tumours were used to test the experimental therapy. Following Omomyc gene treatment, the mice were fed the chemical to turn on the Omomyc gene for four weeks, followed by a four week rest period. All mice became tumour free after the first four week period, but cancers returned in six out of ten mice. After a second four week period of being fed the Omomyc-activating chemical, only one in 10 tumours reappeared. This showed that the gene therapy was very effective at stopping tumour growth.
Cancers are very good at finding ways around treatments and becoming resistant. Because the mice were engineered to continuously develop tumours, the researchers expected them to eventually become resistant and succumb to the disease. However, despite continuous four week cycles of treatment for a year, approximately half the lifespan of a mouse, the cancers did not become resistant. Finally, only two remaining tumours were found after more than a year of treatment among the mice that received eight treatment cycles. Even after such a long periods of treatment, the researchers did not observe any serious side-effects.
The gene therapy used on the mice is not in itself suitable for use in humans, but the experiments show that targeting Myc in this way to selectively knock-out its tumour-forming activities is a viable and very promising approach to beating cancer without the risks of side-effects or resistance. And because Myc is instrumental in so many different cancers, these findings could underpin a range of treatments for patients. The researchers are now working on developing a treatment that will selectively inactivate Myc in humans.
Last edited: 29 July 2022 10:59